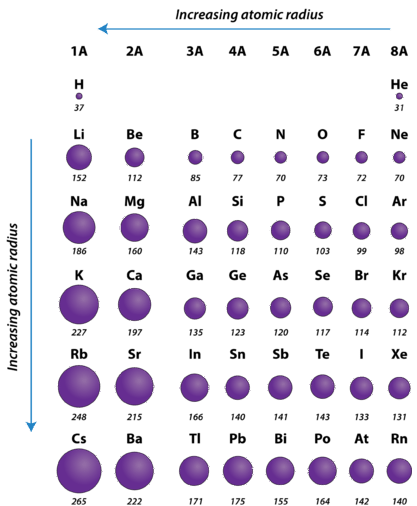 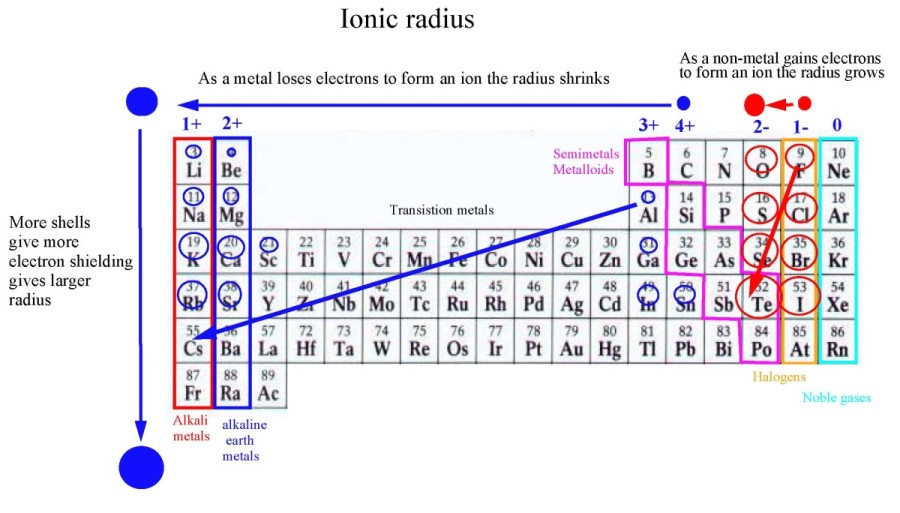
Atomic radius increases going from top to bottom and decreases going across the periodic table. Atomic is the distance away from the nucleus. What is atomic radii and ionic radii?Ītomic and ionic radii are distances away from the nucleus or central atom that have different periodic trends. As a result, the atomic radius decreases. The valence electrons are held closer towards the nucleus of the atom. … This means that the nucleus attracts the electrons more strongly, pulling the atom’s shell closer to the nucleus. Why atomic radii decrease from left to right?Ītomic size gradually decreases from left to right across a period of elements. Opposite charges attract so the negative charge of the electrons is attracted towards the nucleus. … This occurs because as atomic number increases, so do the number of protons, which are positively charged. Why atomic radii do not increase uniformly with increasing atomic number?Ītomic radii decrease in size across the row of the periodic table (as the atomic number increases). Since the force of attraction between nuclei and electrons increases, the size of the atoms decreases. These electrons are gradually pulled closer to the nucleus because of its increased positive charge. Within a period, protons are added to the nucleus as electrons are being added to the same principal energy level. 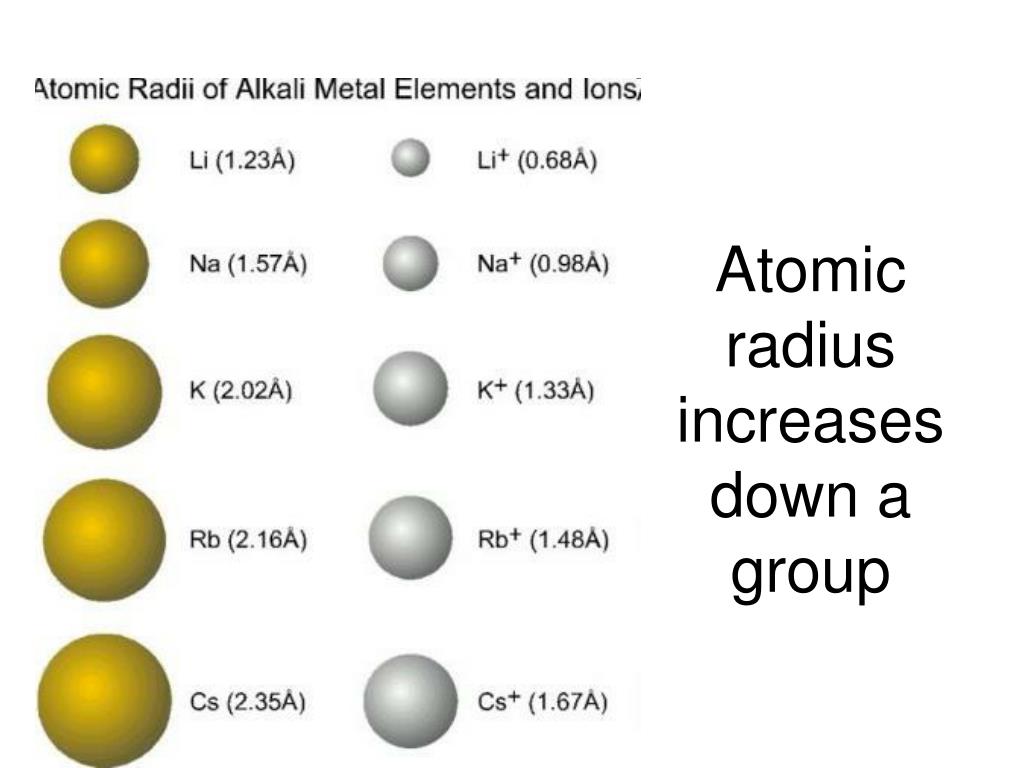
Why does atomic radii decrease across a period? The gain of an electron adds more electrons to the outermost shell which increases the radius because there are now more electrons further away from the nucleus and there are more electrons to pull towards the nucleus so the pull becomes slightly weaker than of the neutral atom and causes an increase in atomic radius. … But for the nonmetallic elements, the ionic radius increases because there are more electrons than protons. Ionic radius decreases moving from left to right across a row or period. This is because each row adds a new electron shell. Why atomic and ionic radii increase down the group?Īs you move down a column or group, the ionic radius increases. Thus, the distance between the nucleus and the last electron will increase and hence, the atomic radius increases down a group. … So, the attraction of the nucleus for the outermost shell electron also goes on decreasing. The electrons get added in new shells which will screen the nucleus. How does atomic size vary down the group and why?.What is the periodic trend observed in the variation of atomic radii down a group?.Why does the change for the atomic radii of the elements in Period 3 from sodium to argon?.What trend is observed among the atomic radii of main group elements down a group?.How does atomic size change with the increase in atomic number?.Why atomic radii decrease from Li to F?.How do atomic radii vary in a period and in a group explain?.Which of the following best explains why the radii decrease from left to right across a period?.Which radii reflect an increase in size of an atom?.How do atomic and ionic radii vary with increasing atomic number?.How does variation of atomic radii in group?.Why does atomic size increase down a group class 10?.Why does the atomic radius increase down Group 2?.Why does atomic radius increase down the group Brainly?.What is the difference between ionic radii and atomic radii?.Why atomic radii decrease from left to right?.Why atomic radii do not increase uniformly with increasing atomic number?.Why does atomic radii decrease across a period?.Why atomic and ionic radii increase down the group?.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
